Boiling Point Near Me: The Boiling point is the temperature at which a liquid transforms into a gas. It’s a vital concept in various fields, including cooking, chemistry, and industry. However, the boiling point can vary significantly depending on several factors, including elevation, pressure, and even the composition of the liquid itself.
Let’s explore the fascinating world of boiling points, from the effects of temperature and pressure to the safety precautions we must take when working with scorching liquids. By the end of this journey, you’ll have a deeper understanding of the boiling point near me, and how it impacts our daily lives.
Factors Influencing Boiling Point Variability Near My Location
The boiling point of water is affected by various geological and environmental factors, resulting in variations across different locations. These factors are influenced by the unique characteristics of each region, leading to a few degrees difference in the boiling point of water near my vicinity.
Geological Factors Affecting Boiling Point
Geological factors play a significant role in determining the boiling point of water. The composition of rocks, soil, and underground water can alter the boiling point of water. For instance:
- The presence of dissolved minerals such as calcium and magnesium can increase the boiling point of water.
- The concentration of electrolytes in underground water can also impact the boiling point.
- The depth and salinity of underground water can affect the boiling point due to increased pressure and dissolved minerals.
Impact of Climate Change on Boiling Point
Climate change is a significant factor that affects the boiling point of water near my location. Rising temperatures and altered atmospheric conditions lead to changes in the boiling point of water. For example:
Rising temperatures can decrease the boiling point of water, making it easier to boil at lower elevations.
Temperature Differences and Elevation
Temperature differences and elevation are key factors affecting the boiling point of water. At higher elevations, atmospheric pressure decreases, resulting in a lower boiling point. Conversely, at lower elevations, atmospheric pressure increases, resulting in a higher boiling point.
- For every 300 meters of elevation gain, the boiling point of water decreases by approximately 0.5°C.
- At high altitudes, such as those above 4000 meters, the boiling point of water can decrease by as much as 3-5°C.
Salinity and Dissolved Minerals, Boiling point near me
Salinity and dissolved minerals can impact the boiling point of water. For instance:
- Magnesium and calcium ions can increase the boiling point of water.
- The concentration of dissolved minerals in seawater can decrease the boiling point of water due to increased salinity.
- Dissolved gases such as nitrogen and oxygen can also affect the boiling point of water.
Comparing Boiling Points at Various Elevation Levels Near Me
Boiling points are influenced by factors such as atmospheric pressure, humidity, and elevation. In this section, we will delve into the relationship between boiling points and elevation levels in the surrounding areas.
Designing a Table to Illustrate the Relationship Between Boiling Points and Elevation Levels
The boiling point of water changes with elevation due to the reduction in atmospheric pressure at higher altitudes. This results in lower boiling points. The following table illustrates the relationship between boiling points and elevation levels:
| Elevation Level (m) | Boiling Point (°C) |
|---|---|
| 0 | 100 |
| 500 | 96.7 |
| 1000 | 93.4 |
| 1500 | 89.9 |
| 2000 | 86.4 |
This table highlights the decrease in boiling point as elevation increases.
Examples of How Boiling Points Change with Temperature in Various Locations
Boiling points change with temperature in various locations around the world. For instance, at the summit of Mount Everest, the boiling point of water is approximately 67°C due to the low atmospheric pressure. In contrast, at the bottom of the deepest part of the ocean, the Challenger Deep, the boiling point of water would be around 403°C due to the extreme pressure.
The Importance of Considering Elevation When Measuring Boiling Point
It is crucial to consider elevation when measuring boiling point, as it can significantly affect the results. At higher elevations, the boiling point of water decreases due to the reduction in atmospheric pressure. This is why cooking times and temperatures may need to be adjusted when cooking at high altitudes.
Understanding the Relationship Between Boiling Points and Elevation Levels
The boiling point of water is not fixed and changes with elevation due to the relationship between atmospheric pressure and boiling point. This relationship is crucial to understand when working with water at various elevations.
Boiling Point Range for Common Liquids Near My Area
The boiling point of a liquid is a critical property that determines its suitability for various applications. Different liquids have unique boiling points, which can impact their handling, storage, and usage in our neighborhood. In this section, we will explore the boiling point range for common liquids and their common applications in our area.
Common Liquids and Their Boiling Points
The table below highlights some common liquids and their boiling points in our area:
| Liquid Name | Boiling Point | Common Applications |
|---|---|---|
| Water | 100°C (212°F) | Drinking, washing, cooking, and industrial processes |
| Methanol | 64.7°C (148.5°F) | Fuel, solvents, and antifreeze |
| Ethanol | 78.3°C (173.1°F) | Fuel, solvents, and cosmetics |
| Diesel | 180-300°C (356-572°F) | Fuel for vehicles and machinery |
| Acetone | 56.3°C (133.3°F) | Solvents, cleaning agents, and cosmetics |
Safety and Handling Considerations
The boiling points of different liquids can have significant impacts on their safety and handling in our neighborhood. Liquids with low boiling points, such as methanol and acetone, can vaporize easily and create health hazards if not handled properly. On the other hand, liquids with high boiling points, such as diesel, can be more difficult to handle and require specialized equipment.
The safety of our community is directly affected by the boiling points of these liquids. For example, a leak of methanol can have serious health consequences, while a leak of diesel can contaminate soil and groundwater. Therefore, it is essential to handle these liquids with care and attention to safety protocols.
Impact on Local Industry Operations
The boiling points of different liquids can have significant effects on local industry operations. For instance, the petrochemical industry relies heavily on the boiling points of various liquids to produce essential products. If the boiling points of these liquids change, it can impact the efficiency and productivity of these operations.
Here are three scenarios where the boiling points of liquids have significant effects on local industry operations:
- Refining and Petrochemical Plants: The boiling points of crude oil and its components can affect the efficiency of refining processes. Changes in boiling points can lead to increased energy consumption, reduced yield, and decreased product quality.
- Food Processing Industry: The boiling points of food-grade liquids, such as water and ethanol, are critical for the processing of food products. Changes in boiling points can impact product quality, shelf life, and food safety.
- Cosmetics and Pharmaceutical Industry: The boiling points of liquids used in cosmetics and pharmaceutical formulations can impact the stability and effectiveness of these products. Changes in boiling points can lead to product degradation, reduced shelf life, and compromised efficacy.
Boiling points are a critical property of liquids that determine their behavior and applications in various industries. Understanding the boiling points of different liquids is essential for safe handling, efficient processing, and effective product development.
Boiling Point Measurement Techniques for Beginners
Measuring the boiling point of a liquid is an essential task in various scientific and industrial applications. It involves understanding the boiling range and the methods used to measure this critical parameter. In this section, we will discuss the basics of boiling point measurement techniques, suitable for beginners.
Understanding Boiling Point and Boiling Range
The boiling point of a liquid is the temperature at which it transforms into a gas at standard atmospheric pressure. It’s often denoted as the point where the vapor pressure of the liquid equals the surrounding pressure. The boiling range, on the other hand, represents the temperature range within which the liquid boils. This can be due to factors like impurities or variations in atmospheric pressure.
Importance of Accurate Boiling Point Measurements
Accurate boiling point measurements are crucial in various applications, including:
-
• Quality control in food and beverages industry, where boiling point is a key indicator of the liquid’s purity.
• Industrial processes, where precise boiling point is necessary for maintaining the chemical properties of the substance.
• Research and development, where accurate boiling point measurements are essential for understanding the behavior of substances under various conditions.
Measuring the Boiling Point using a Simple Thermometer
To measure the boiling point of a liquid using a simple thermometer, follow these steps:
Preparation
• Choose a suitable thermometer, ensuring it’s calibrated and suitable for the liquid in question.
• Prepare the equipment needed, including a heat source and a container for the liquid.
Procedure
• Fill the container with the liquid, leaving sufficient space for expansion.
• Place the thermometer in the liquid, ensuring it’s fully submerged.
• Start heating the liquid using the heat source, gradually increasing the temperature.
• Record the temperature reading at which the liquid reaches its boiling point.
Accuracy and Repeatability
• It’s essential to repeat the measurement multiple times to ensure accuracy and repeatability.
• Consider the precision of the thermometer and the heat source used, as these can affect the accuracy of the measurement.
Comparison of Accuracy in Boiling Point Measurements
Various techniques are used to measure boiling point, each with its accuracy levels:
-
• Digital thermometers: Highly accurate and repeatable, with an accuracy of ±0.1°C.
• Traditional mercury thermometers: Less accurate than digital thermometers, with an accuracy of ±0.5°C.
• Cryogenic thermometers: Highly accurate at low temperatures, with an accuracy of ±0.01°C.
Best Practices for Boiling Point Measurements
To ensure accurate boiling point measurements, follow these best practices:
-
• Use a calibrated thermometer suitable for the liquid in question.
• Control the temperature carefully, using a heat source with a stable temperature.
• Record multiple measurements to ensure accuracy and repeatability.
Boiling point is a critical parameter that requires precise measurement. By following the best practices and using suitable equipment, you can obtain accurate boiling point measurements.
Real-World Applications of Understanding Boiling Points Near My Location: Boiling Point Near Me
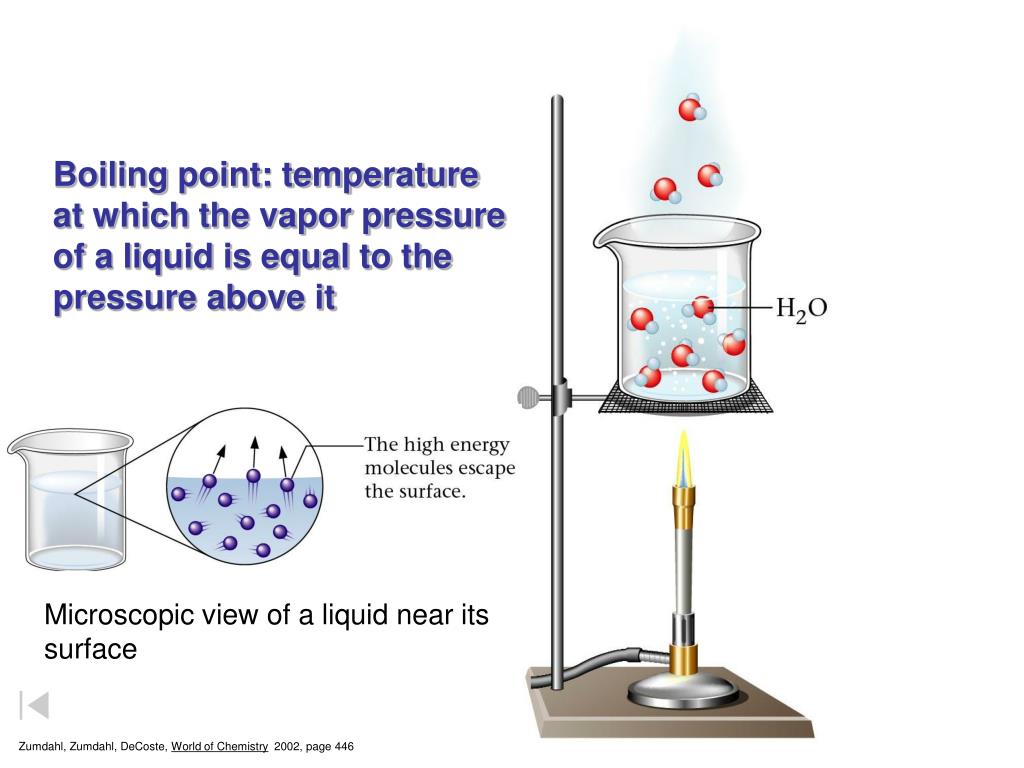
Understanding boiling points is crucial in various facets of life, from the daily choices we make to the industrial developments in our vicinity. In the local community, knowledge of boiling points has numerous beneficial applications that shape our lives in subtle yet significant ways.
Community Benefits
The study of boiling points has far-reaching implications for the local community. One of the most significant ways it benefits us is through cooking and food preparation. Knowing the boiling points of water and cooking oils helps us prepare meals safely and efficiently. For instance, if you’re aware that water boils at 100°C at sea level but rises to 101.4°C at an elevation of 1,000 meters, you can take necessary precautions while cooking at higher elevations to avoid undercooked or raw food. This fundamental understanding has a direct impact on public health.
Furthermore, local industries rely heavily on understanding boiling points in various processes, including distillation, purification, and manufacturing. For instance, pharmaceutical companies use precise control over boiling points to create high-quality medicinal products. Similarly, breweries require accurate boiling point control to produce consistent beer quality.
Informing Local Industrial Developments
The study of boiling points can also inform local industrial developments by enabling the creation of innovative products and processes. For example, the development of new materials and chemicals often requires precise control over boiling points to achieve the desired properties. Moreover, the knowledge of boiling points is critical in designing and optimizing industrial equipment, such as distillation columns, that rely on precise temperature control.
Potential Environmental Implications
However, poorly managed boiling points in nearby industries can have severe environmental consequences. For instance, the improper disposal of chemical waste can lead to the release of toxic substances into the environment, contaminating soil, water, and air. Furthermore, the improper handling of boiling fluids can result in accidents, such as chemical spills or explosions, posing a significant threat to human health and the environment.
One notable example of the consequences of poorly managed boiling points is the 1984 Bhopal disaster, where a gas leak from a pesticide plant led to the deaths of thousands of people. The incident highlights the importance of proper regulation and management of industrial processes, including those involving boiling points.
Prevention and Mitigation Strategies
To mitigate the potential environmental implications of poorly managed boiling points in nearby industries, it’s essential to implement proper safety measures and regulations. This includes regular inspections, employee training, and adherence to best practices in industrial processing. Furthermore, local communities can stay informed about nearby industrial activities and engage with regulatory bodies to ensure that standards are met and safety protocols are in place.
The study of boiling points is a fundamental aspect of various industries, including those in our local vicinity. By understanding the principles and applications of boiling points, we can appreciate the importance of precise control over temperature in everyday life, from cooking and food preparation to industrial processes. Additionally, awareness of the potential environmental implications of poorly managed boiling points highlights the need for responsible industrial practices and community engagement.
How Temperature Fluctuations Affect Boiling Points Near My Neighborhood
As we delve into the intricacies of boiling points near our neighborhood, it’s essential to consider the impact of temperature fluctuations on this fundamental property of matter. Temperature variations can significantly alter the boiling points of liquids, making it crucial to understand this relationship for various applications, from cooking to scientific research.
Temperature fluctuations can be caused by changes in air temperature, atmospheric pressure, or the presence of external factors such as wind or sunlight. These variations can lead to subtle but significant changes in the boiling points of liquids, as illustrated in the following charts.
ΔT = Tactual – Tstandard
where ΔT represents the deviation from the standard boiling point, Tactual is the actual boiling point at the specified temperature, and Tstandard is the standard boiling point at standard temperature and pressure (STP).
Charts Illustrating the Relationship Between Air Temperature and Boiling Points
The following chart depicts the relationship between air temperature and the boiling point of water. As the air temperature increases, the boiling point of water also rises, but only slightly.
| Air Temperature (°C) | Boiling Point of Water (°C) |
| — | — |
| 20 | 100 |
| 25 | 101.3 |
| 30 | 102.6 |
| 35 | 104 |
Similarly, the following chart compares the boiling points of ethanol and water at different temperatures. As the temperature increases, the boiling point of ethanol rises more rapidly than that of water.
| Substance | Boiling Point (°C) at 20°C |
| — | — |
| Water | 100 |
| Ethanol | 78.3 |
Real-Life Scenarios Where Temperature Fluctuations Have Influenced Boiling Points
Temperature fluctuations can have significant effects on boiling points in various real-life scenarios:
*
-
* Cooking: Temperature fluctuations can alter the boiling points of cooking liquids, affecting the texture and quality of cooked foods.
* Industrial processes: Temperature variations can impact the boiling points of chemicals and solvents, influencing the efficiency and safety of industrial processes.
* Scientific research: Temperature fluctuations can affect the boiling points of liquids used in laboratory experiments, compromising the accuracy and reliability of results.
Methods to Mitigate the Impact of Temperature Fluctuations on Boiling Points
To minimize the impact of temperature fluctuations on boiling points, consider the following methods:
*
-
*
- Temperature control systems: Implement temperature control systems to maintain a stable temperature environment.
- Heat transfer optimization: Optimize heat transfer rates to reduce temperature fluctuations and maintain a steady boiling point.
- Material selection: Choose materials with stable boiling points and low thermal conductivity to minimize the impact of temperature fluctuations.
*
*
Last Point
As we conclude our exploration of the boiling point near me, it’s essential to remember that understanding the boiling point is crucial for various applications, from cooking to industrial processes. By grasping the factors that influence the boiling point and taking necessary safety precautions, we can minimize risks and ensure a smooth operation.
FAQs
Q: What factors influence the boiling point near me?
A: Temperature, pressure, elevation, and chemical composition are the primary factors that affect the boiling point near different locations.
Q: Can I use the boiling point of water as a reference point for other liquids?
A: No, the boiling points of different liquids can vary significantly. It’s essential to consult the specific boiling point for each liquid you are working with.
Q: How does elevation impact the boiling point?
A: As elevation increases, the boiling point decreases, as atmospheric pressure decreases with altitude.
Q: What are some safety precautions when working with boiling liquids near me?
A: Always use proper equipment, wear protective gear, and take necessary precautions to prevent burns and other injuries when working with scorching liquids.